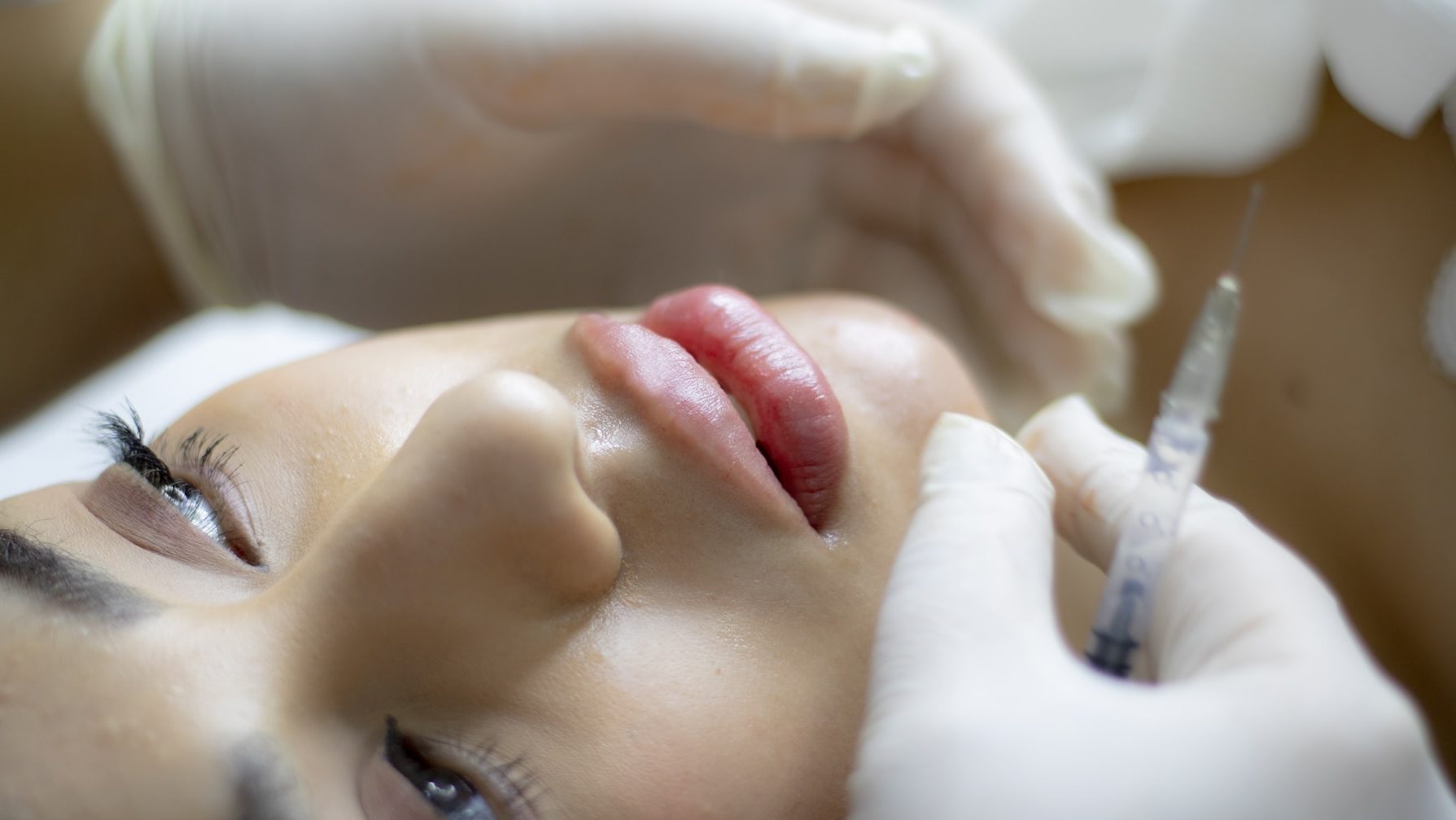
Facial Fillers as Medical Devices
Facial Fillers as a Medical Device
Amy Eikelenboom
Co-founder of NAALA
Published on 10 September 2021
Do you manufacture facial fillers? You might be developing a medical device. The industry of facial or other dermal fillers have faced several regulatory changes over the year. Historically, dermal fillers have been marked as medical devices, medicinal product, or cosmetics. With the applicability of the European Medical Devices Regulation (hereinafter: “EU MDR”) last May, the EU has tightened the requirements applicable to facial fillers. Following Annex XVI, facial fillers qualify as medical devices.
But facial fillers do not have a medical purpose? Why are they a medical device?
That is true. Facial fillers do not have a medical purpose, as defined by the EU MDR [1]. However, the EU MDR extends the legislative scope to aesthetic products without an intended medical purpose.
Annex XVI refers to facial fillers as follows[1]:
Substances, combinations of substances, or items intended to be used for facial or other dermal or mucous membrane filling by subcutaneous, submucous or intradermal injection or other introduction, excluding those for tattooing.
The types of devices included are perceived to have the same characteristics and risk profile as medical devices.
Why do facial fillers have a high-risk profile? That is determined by the following two characteristics of Facial fillers:
- they achieve their intended use by biophysical properties rather than through pharmaceutical, metabolic, or immunological actions, and
- they are injected into the human body, which has a high-risk profile.
How are facial fillers classified?
Facial fillers will most likely be inside the body for more than 30 days. The EU MDR defines this as ‘long term use’[1]. Additionally, facial fillers are most likely to be wholly or mainly absorbed into the human body. According to the classification rules of the EU MDR, they will most likely be a class III medical device (the highest possible risk class).[2]
What requirements apply for facial fillers?
When classifying as a class III medical device, facial fillers are subject to the most strict set of requirements applicable to a medical device.
Pursuant to the type of conformity assessment [3], the following should be in place at minimum:
- Establish, document and implement a quality management system, commonly referred to as QMS. A QMS is the internal system to ensure your device is manufactured and delivered meeting your defined policies, procedures and records. The European Commission has identified ISO 13485 as a harmonized standard, which may be used to give substance to this requirement;
- Establish and update technical documentation describing the underlying architecture, design material, manufacture, interfacing process and performance of the medical device in question;
- Obtain sufficient clinical data by performing a clinical evaluation and, when applicable a clinical investigation [4]. A clinical evaluation is an ongoing procedure to collect, appraise and analyze clinical data to evaluate whether there is sufficient clinical evidence to confirm compliance. When a clinical investigation is required, an investigation in one or more human subjects must be undertaken to assess the safety or performance of the device in question.
- Provide the implant card [5]. An implant card contains the name of the device, the serial number and/or lot number, the UDI, the name, address and website of the manufacturer and the device type (model). The implant card is aimed to enable patients to identify the device;
- Provide a summary of safety and clinical performance [6]. A summary of safety and clinical performance is intended to provide public access to an updated summary of clinical data and other relevant information about the safety and clinical performance of the device;
- Provide a Periodic Safety Update Report [7]. A Periodic Safety Update Report, commonly referred to as PSUR, presents the (re)evaluation of the risk-benefit balance of the device;
- Meet common specification for products without a medical purpose. Common Specifications are technical or clinical requirements provided by the European Commission to give substance to legal obligations applicable to the device. The common specifications for facial fillers are expected in Q3 of 2021 according to the implementation rolling plan of the European Commission [8];
- Implement Post-market surveillance activities. These activities are aimed to proactively collect and review real-life experiences with the device and to apply necessary corrective or preventive actions;
- Partner with designated notified body to undergo the conformity assessment in order to obtain the CE mark [2];
- In case you are not physically present in the European Union, appoint a European Authorized Representative. The European authorized representative will act on your behalf in carrying out certain tasks;
- Etc.
Manufacturers that have a valid CE mark obtained under the Medical Device Directive (the predecessor of the EU MDR), may be subject to the transition period. All other manufacturers that fail to meet EU MDR requirement, may not market their products in the European Union.
NAALA is specialized in supporting manufacturers with meeting EU MDR requirements in order to obtain the CE mark under the MDR. Want to know how we can support you specifically? Feel free to reach out.
[1] Consolidated TEXT: 32017R0745 — EN — 05.05.2017 (europa.eu)
[2] Classification rule 8, EU MDR
[3] Facial filler may follow the conformity assessment specified in Annex IX or a combination of the conformity assessment procedures, specified in Annex X and Annex XI,
[4] MEDDEV 2.7/1 revision 4 may be useful to structure your clinical evaluation
[5] Article 18, EU MDR
[6] Article 32, EU MDR
[7] Article 86, EU MDR
Please note that all details and listings do not claim to be complete, are without guarantee and are for information purposes only. Changes in legal or regulatory requirements may occur at short notice, which we cannot reflect on a daily basis.
Other articles you may be interested in:
Liked the article? Maybe others will too. Feel free to share!